GAV 2.0
FEATURES
- ONE valve for premature and newborn babies, children and adults
- three product variants: for ventriculo-peritoneal (GAV 2.0) and lumbo-peritoneal drainage (GAV 2.0 LP, straight and GAV 2.0 LP, U-shaped)
- more protection against overdrainage complications due to position-dependent function
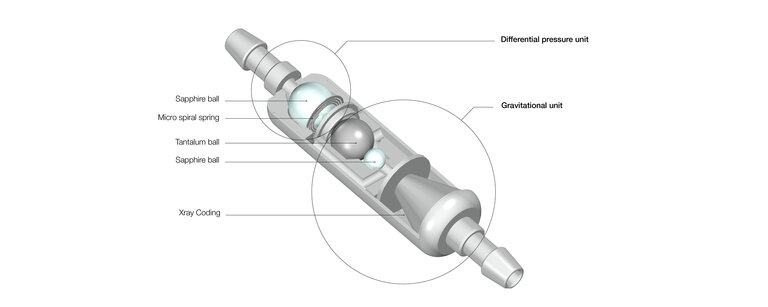
- uncomplicated pressure stage concept with 6 different fixed pressure stages to choose from
- simple pressure level coding for clear pressure level detection in imaging
- MRT compatible up to 3 Tesla
- slim, cylindrical and small design for a better flow
- precise precision engineering
robust and durable due to the use of titanium
GAV 2.0
FUNCTIONALITY
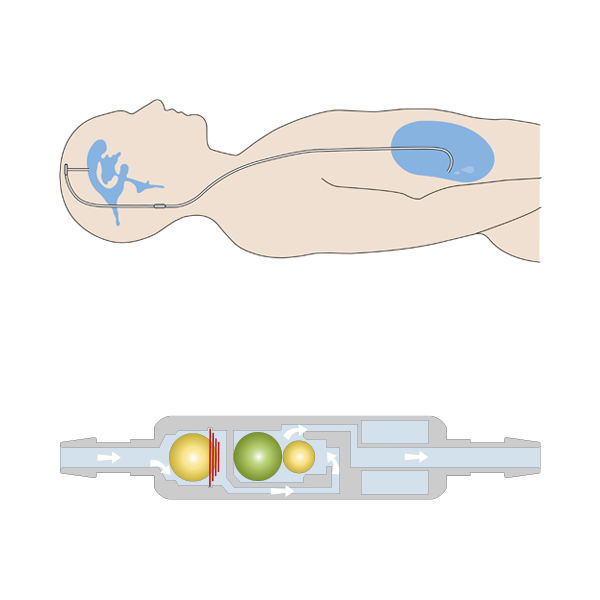
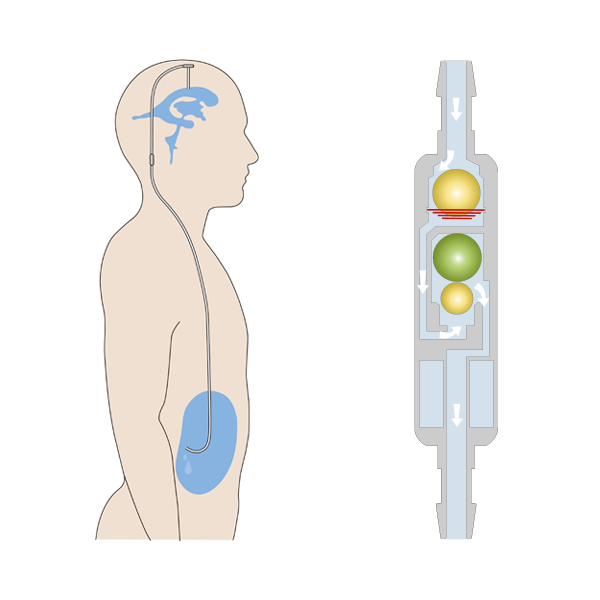
The GAV 2.0 must always be implanted parallel to the body axis for optimal function. It then automatically adapts its opening pressure to the different body positions of the patient and can thus meet the different requirements of a shunt system in lying and standing positions and, above all, counteract the risk of possible overdrainage in the upright body position.
One valve for children and adults
The slim cylindrical design of its predecessor has been further optimised and further reduced in the GAV 2.0 for a better flow pattern. Coupled with the modified pressure stage concept, the GAV 2.0 is suitable for the treatment of hydrocephalus in premature and newborn babies, children and adults.
PRESSURE LEVEL DETECTION IN X-RAY IMAGE
For detailed information on the optimum pressure stage setting, please refer to our pressure stage recommendation.

MIETHKE
Valve Board
Our Valve Boards

OUR MIETHKE PRODUCT FINDER
We consider the individual patient situation, surgical technique and treatment plan. The questions are designed in such a way that the list of results provides a maximum of three product recommendations.
DO YOU HAVE ANY QUESTIONS ABOUT THE PRODUCT?
WE ARE THERE FOR YOU
![[Translate to English:] Zwei Marketing Mitrabeiter*innen blättern durch M.scio Prospekte von MIETHKE](/fileadmin/_processed_/f/8/csm_SalesTeam_a8fc997cce.png)
FOR OUR SALES EXPERTS
We are there for you!
Dear sales & shunt experts all over the world,
our toolbox is big. If you need something to make MIETHKE shunts easier to understand or have new ideas, please get in touch with us. We look forward to exchanging ideas and - who knows - maybe what you are looking for is already there and we can easily make it available to you. Or we can develop something with you that could also be very helpful for other markets.
Your MIETHKE Marketing Team

OUR PARTNERSHIP
WITH B. BRAUN
B. Braun and MIETHKE - Together for a better life with hydrocephalus
We have a long and intensive partnership with B. Braun in the field of neurosurgery. We are driven by a common vision: to improve the lives of hydrocephalus patients around the world with innovative solutions.
Our partnership is an exciting combination of B. Braun's many years of expertise as one of the world's leading medical device and pharmaceutical companies and our agility as an innovative company and technology leader in gravitation-based shunt technology.
Our Strong Partner in Neurosurgery: